Zeta Potential II: cmPALS technology, Omega Cuvette design, and practical guidance for reliable measurements
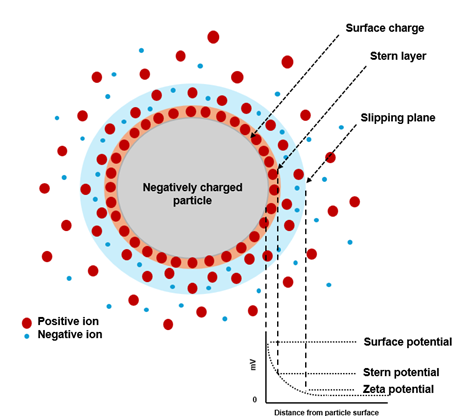
Zeta potential is a key parameter for characterizing the stability and interparticle interactions of colloidal dispersions, nanoparticles, emulsions, and biological systems. In simple terms, it describes how strongly particles in a liquid repel or attract each other and therefore whether a dispersion tends to stay stable or to aggregate. Particles dispersed in a liquid typically carry an electrical charge on their surface, arising from ionization, adsorption of ions, or surface functional groups (Figure 1).
This article builds on "Zeta Potential I: Principles" and focuses on how the cmPALS technique and Anton Paar's cuvette designs turn these concepts into fast, reliable, and easy-to-perform measurements in practice. For beginners, this section explains the basic steps and choices needed to obtain trustworthy zeta potential results. For experienced users, it highlights how advanced features such as cmPALS and the Omega cuvette improve data quality, reduce sample stress, and extend cuvette lifetime.
From ELS to cmPALS: Why the measurement principle matters
When an external electric field is applied, charged particles migrate toward the oppositely charged electrode. The velocity of this motion, known as electrophoretic mobility, can be related to the zeta potential, which corresponds to the electrical potential at the slipping plane surrounding the moving particle. Because zeta potential reflects how strongly particles repel or attract each other, it provides direct insight into dispersion stability, aggregation tendencies, and surface chemistry.
To measure electrophoretic mobility with high sensitivity and accuracy, modern ELS instruments employ continuously monitored Phase Analysis Light Scattering (cmPALS). With cmPALS, the phase shift of the scattered light is monitored continuously while only short, low-intensity electric field pulses are applied, so the instrument can detect very small particle movements without stressing the sample. An additional detector continuously measures the Doppler shift of the modulated reference beam, which compensates for instabilities in the modulator and enables precise phase analysis even at low voltages. This greatly reduces sample heating and electrochemical reactions at the electrodes, making it particularly suitable for delicate, low-conductivity, or highly concentrated samples.
Compared to classical electrophoretic light scattering approaches, cmPALS enables faster zeta potential measurements, improved repeatability and sensitivity, and reliable results even under challenging conditions.
Working with cmPALS means that you can rely on the default settings to obtain robust zeta potential values without having to optimize voltage or measurement time for every sample. The high sensitivity at low field strengths allows reliable measurements for delicate, low-conductivity, or concentrated samples that are often problematic on conventional ELS instruments, while at the same time reducing electrode aging and cuvette fouling.

Measurement cells: Understanding cuvette design in zeta potential measurement
For zeta potential measurements, there are typically two types of cuvettes: disposable cells and zeta potential cells that are immersed in the sample.
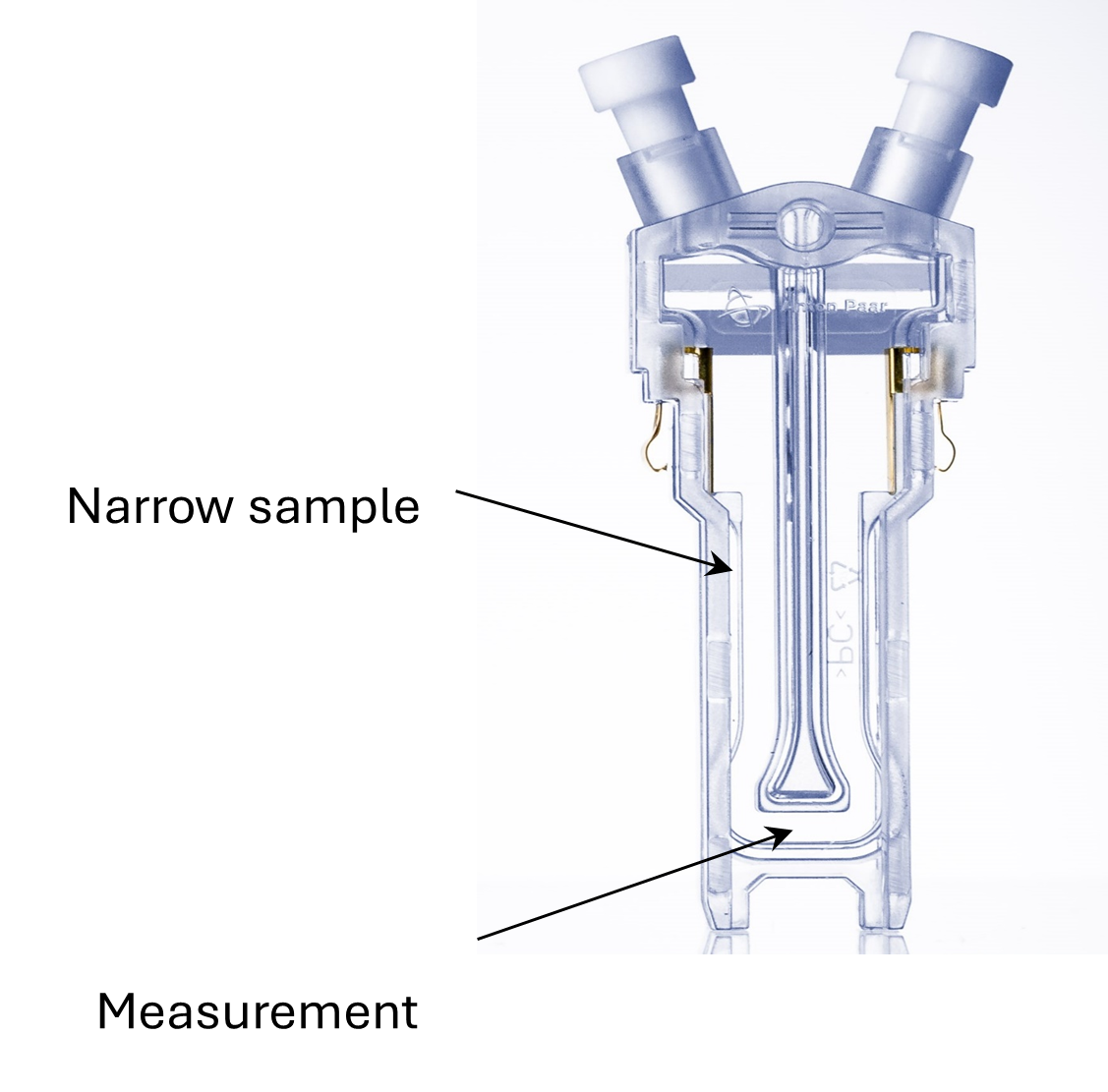
Disposable cells, such as Anton Paar's patented Omega cuvette, are ideal for routine measurements. Made from durable polycarbonate, the Omega cuvette offers chemical stability as well as high abrasion and scratch resistance. It contains two gold-coated electrodes inside a narrow capillary channel, which is shaped like an inverted omega instead of a conventional U-shape. Its standout feature is the distinctive geometry of the capillary tube, which prevents the formation of electric field-strength gradients in the section of the capillary where particle velocity is measured, ensuring more reliable and stable results.
Straight to the advantage: The inverted Omega
In conventional ELS cuvettes, zeta potential is typically measured at the bottom of the tube – an area that is often U-shaped and curved in many disposable zeta potential cells. This U-shape introduces a key limitation: the electric field in curved sections is not uniform. Because electric current always prefers the shortest path, most of the current in a U-shaped cuvette flows along the inner wall, creating strong field gradients and position-dependent zeta potential values. As a result, the measured zeta potential can vary significantly depending on the exact measurement position within the cuvette.
To overcome this challenge, Anton Paar developed an innovative zeta potential cell in which the sample capillary is not U-shaped, but instead follows an inverted-omega design, as shown in Figure 3. Unlike the traditional U-shaped geometry, the omega-shaped capillary features a long, straight section at its lowest point. This straight segment ensures a homogeneous electric field in the region where the measurement is performed, preventing the formation of electric-field gradients. Consequently, measurements performed with Anton Paar's Omega cuvettes are highly stable, reproducible, and independent of the lateral measurement position across the capillary.
Simulations further highlight this advantage. In the Omega cuvette, the electric field remains nearly constant across the cross-section (Figure 4, dark blue line), whereas in a U-shaped cuvette, the field strength varies substantially (Figure 4, red line).
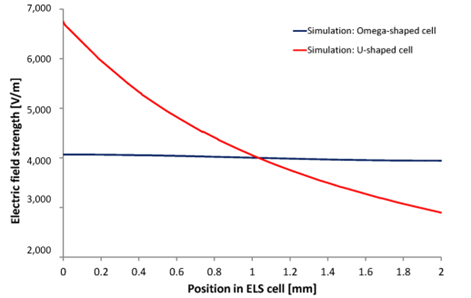
This difference arises from the fundamental behavior of electric current, which naturally follows the shortest available path. In a U-shaped cuvette, the shortest route lies close to the upper wall. As a result, most of the current concentrates in this region, with substantially less flowing near the lower wall. Consequently, the locally experienced electric field, and thus the measured zeta potential, varies depending on the measurement position. Accurate results can only be obtained if the measurement is taken exactly at the center of the channel, making the setup highly sensitive to alignment.
In contrast, simulations of the Omega-shaped cuvette reveal that the electric current is distributed uniformly across the measurement cross-section. This homogeneous current distribution ensures that the zeta potential remains essentially independent of the lateral measurement position.
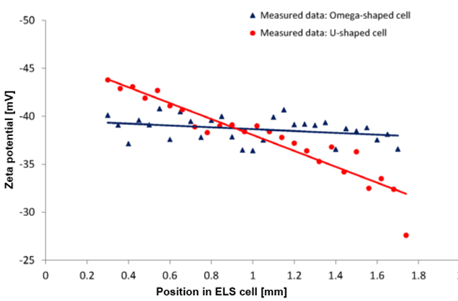
The experimental data in Figure 5 strongly support these predictions: In the U-shaped cuvette, the zeta potential measured near the upper wall is approximately 44 mV, nearly 40 % higher than the value measured near the outer wall (around 32 mV). This clearly illustrates the presence of an electric-field gradient across the capillary. In the Omega cuvette, however, the zeta potential remains nearly constant across the channel (39 mV at the upper wall and 38 mV at the lower wall), confirming that the applied field is homogeneous. This uniformity greatly reduces sensitivity to positioning errors and ensures highly reliable measurement results.
Beginners do not need to worry about precisely hitting the "correct spot" in the cuvette, as the Omega geometry ensures comparable results every time. For experienced users, the homogeneous field and reduced sensitivity to alignment translate into superior reproducibility, lower method-development effort, and more robust comparison between different instruments and laboratories.
Choosing between the Omega cuvette and the Univette in practice
For most routine aqueous dispersions, nanoparticle suspensions, and biological formulations where convenience and throughput are paramount, the disposable Omega cuvette is the preferred choice, as it minimizes preparation effort, avoids cross-contamination, and provides excellent repeatability thanks to its well-defined field geometry and electrode design. Its design supports low-sample volumes and is well-suited for low-conductivity buffers that benefit most from using low-voltage cmPALS.
The Univette is a reusable cuvette for dynamic and electrophoretic light-scattering measurements. Designed for broad applicability, it is compatible with both aqueous and organic solvents and supports a wide range of sample conditions, including high-concentration dispersions, low sample volumes, and elevated temperatures. The Univette is made of PEEK and equipped with palladium electrodes, which can easily be cleaned. Its reusability reduces consumable demand while maintaining reliable performance.

In practice, many laboratories use Omega cuvettes for everyday aqueous work and Univette cells for method development, organic dispersions, or conditions that would otherwise shorten the lifetime of disposable electrodes.
Conductivity in typical DLS and zeta potential measurement applications
The conductivity of the sample strongly influences zeta potential measurements because high ionic strength increases Joule heating and electrochemical reactions at the electrodes, while also compressing the electric double layer and reducing the absolute zeta potential. For most DLS and electrophoretic light scattering applications, however, the conductivities encountered in practice are far below the upper specification limit of
200 mS/cm used for Litesizer DLS instruments.
Table 1 gives approximate conductivity ranges for common dispersions, solutions, and emulsions encountered in routine zeta potential measurements. While these values are indicative and can vary with exact composition, temperature, and formulation, they illustrate that typical samples used for DLS and cmPALS are usually in the low- to mid-mS/cm range.
Table 1. Approximate conductivities of DLS-relevant samples.
| Sample type | Typical conductivity at 20 °C to 25 °C (mS/cm) |
| Ultrapure water | < 0.1 |
| Deionized water exposed to air | 0.5 to 2 |
| Low-ionic-strength buffer (e.g., 1 mM to 5 mM salt) | 0.5 to 3 |
| Typical DLS buffer (e.g., 10 mM to 50 mM phosphate, HEPES, Tris) | 3 to 15 |
| PBS / physiological saline (~150 mM ionic strength) | 12 to 18 |
| Cell culture media or protein formulations with electrolytes | 10 to 20 |
| Cosmetic oil-in-water emulsions with typical electrolyte content | 1 to 10 |
| High-salt process or cleaning solutions (> 0.5 M salt) | 50 to 150+ |
| Saturated brine or electroplating baths | 150 to 250+ |
As this overview shows, the vast majority of nanoparticle dispersions, biological formulations, and cosmetic emulsions used for zeta potential analysis have conductivities well below 20 mS/cm, and even demanding high-salt media typically remain far below 200 mS/cm.
Conductivities far beyond this region are mainly observed in extremely concentrated industrial electrolytes, which are rarely measured by DLS due to strong multiple scattering, increased sample heating, and limited interpretability of zeta potential at very short Debye lengths.
Challenges for zeta potential measurement: Electrode fouling and cuvette lifetime
During zeta potential measurements, electrode surfaces can undergo electrochemical reactions that lead to the formation of dark deposits, most commonly a black or brown film, which is shown in Figure 7.

These deposits originate from oxidation or reduction of sample constituents, such as organic additives, polymers, surfactants, or metal ions, or from carbonization of residual material at the electrode surface. Persistent fouling can impair the stability of the applied electric field, reduce signal quality, and ultimately compromise measurement accuracy.
Regular visual inspection of the electrodes combined with measurements of a well-characterized zeta potential control standard is therefore the most reliable way to assess whether a cuvette is still suitable for use. The usability of a cuvette can be assessed through visual inspection of the electrodes and comparison with a well-characterized reference sample. A cuvette should be considered unsuitable when discoloration or deposits cannot be removed by standard cleaning procedures, when the electrode surface shows signs of corrosion or pitting, or when measurements display excessive noise, poor repeatability, or deviations from established reference values.
In practice, a cuvette should be replaced when (1) deposits cannot be removed by standard cleaning, (2) corrosion or pitting of the electrodes becomes visible, or (3) a control standard starts to show increased noise, poor repeatability, or values outside the specified validation range.
Long-term tests with Anton Paar's zeta potential control have shown that an Omega cuvette can deliver more than 1,000 valid measurements under typical conditions, even if the electrodes appear darkened, as long as the reference values remain within the predefined limits.
To make things even more complex, there are cases where the electrodes appear black, yet the measurements are still valid. Figure 8 shows that after 330 measurements, the zeta potential values were already close to the lower limit of the valid range for the zeta potential control standard (Anton Paar). After these measurements, the measurement was stopped, and the cuvette was inspected. The electrodes had darkened after the numerous measurements, and many air bubbles were visible both within the sample and on the electrodes. This suggests that changes in the sample occurred during the measurement.
The cuvette was then refilled with a fresh zeta potential control standard. Upon refilling, a clear step in the results was observed, and the zeta potential values returned to normal. The results after refilling indicated that the measurement cell was still in good condition, despite the blackened electrodes. This procedure was repeated after 400 measurements and 399 measurements. After each refilling, a distinct step in the results was again observed. However, after 1,129 measurements, the mean zeta potential values began to approach the lower validation limit for the zeta potential control, indicating that the cell may have reached the end of its lifetime. In total, 1,308 measurements were performed with the same Omega cuvette.
![Figure 8: Mean zeta potential [mV] over the number of measurements](https://wiki.anton-paar.com/fileadmin/wiki/images/23821_Zeta-Potential-Measurement/Figure-8-Zeta-Potential.png)
To extend the lifetime of Omega and Univette cuvettes in daily work:
- Rinse the cuvette immediately after each measurement with a suitable solvent and avoid abrasive cleaning tools that might damage the electrodes
- Use the lowest electric field and shortest measurement time that still give stable results; cmPALS helps here by providing high sensitivity even at low voltages
- Regularly verify performance with a zeta potential control standard to detect potential drift early and to avoid discarding still-functional cuvettes based on appearance alone
Troubleshooting: Recognizing cuvette-related issues
When zeta potential measurement data become unreliable, it is important to distinguish cuvette-related problems from sample instability or instrument settings. Typical signs of a compromised cuvette include unusually high noise, poor repeatability between consecutive runs, systematic drift in zeta potential for a stable reference, or strong dependence of the result on the measurement position within the cuvette – effects that the homogeneous field of the Omega geometry is specifically designed to suppress.
A practical troubleshooting workflow includes:
- Checking for air bubbles or incomplete filling, as these can distort the local field and scattering signal.
- Verifying that the sample conductivity lies within the recommended range for the chosen cell and settings, since very high conductivity can accelerate electrode reactions. For most samples, a conductivity between 0.1 mS/cm and 1 mS/cm gives reliable results while extending the lifetime of the cuvettes.
- Comparing results with a fresh Omega cuvette or a well-maintained Univette to separate cuvette effects from sample or method issues.
Conclusion
Zeta potential is a powerful indicator of dispersion stability, and accurate zeta potential measurement relies not only on advanced measurement techniques such as cmPALS, but also on the design and condition of the measurement cuvette. With cmPALS, the Omega cuvette, and the reusable Univette, Anton Paar provides a practical toolkit that makes zeta potential measurements accessible for beginners while at the same time offering the sensitivity, robustness, and lifetime expected by experienced users in demanding research and quality-control environments.
Anton Paar's Omega cuvette overcomes the limitations of conventional U-shaped geometries by providing a homogeneous electric field, resulting in highly reliable and position-independent zeta potential measurements. Complementary options such as the reusable Univette further expand the range of suitable sample types and measurement conditions. Proper cuvette handling, regular inspection, and awareness of electrode fouling are essential to maintaining measurement quality. With robust instrumentation, thoughtfully designed cuvettes, and careful laboratory practice, users can achieve consistently accurate zeta potential results across a broad range of applications and analytical challenges.