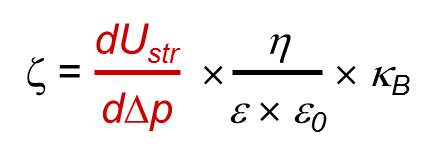Zeta Potential I: Principles
Over the last few decades zeta potential measurements have become an important characterization method for surface functionality or stability of dispersed particles. The main focus of zeta potential analysis is gaining information on the surface charge of a material. The material can range from colloidal nanoparticles up to macroscopic surfaces like membranes or silicon wafers. This article describes the theory behind the zeta potential, explains why it is dependent on the pH value and ionic strength of the medium, and describes the techniques which are used for zeta potential analysis.
What is zeta potential?
The zeta potential (also known as electrokinetic potential) is established on the surface of any material when it comes in contact with a liquid medium. It is thus an interfacial property. It is typically given in millivolt unit.
If a material comes in contact with a liquid, the functional groups on its surface will react with the surrounding medium. This process results in a surface charge, which attracts the accumulation of oppositely charged ions. These counter ions arrange themselves spontaneously in a so-called electrochemical double layer. The zeta potential is defined as the sum of the initial surface charge and the accumulated layer.
The following needs to be kept in mind when talking about zeta potential:
- The zeta potential is only present if a material comes in contact with a liquid. It represents the effective net charge in this condition.
- The zeta potential can be measured on macroscopic surfaces (e.g. membranes, hair, polymers) as well as from particles dispersed in a liquid (e.g. colloids, nanoparticles, liposomes). Both types are considered “material”. However, the differentiation is important in order to choose the right technology to measure the zeta potential.
- The properties of the liquid medium play an important role in the formation of the zeta potential, which is highly dependent on the pH value or buffer concentration.
- A surface charge can be observed for solid materials (solid-liquid interface) as well as liquid droplets (liquid-liquid interface).
Why the zeta potential is important
Measuring the zeta potential provides information on surface functionality, the stability of dispersed particles as well as interaction of dissolved compounds with the solid surface.
The zeta potential of macroscopic surfaces is thus important for understanding the behavior of solid materials in many technical processes in which aqueous systems play a role, e.g. membranes for water treatment, biomaterials in contact with blood, or wet processing of semiconductor wafers. Knowledge of the zeta potential of a material helps you optimize specific surface modification processes for a material to perform at its best when applied.
The zeta potential of particles is a key indicator of the stability of a colloidal dispersion, like nanoparticles or liposomes, since it reflects the ability of particles to repulse each other electrostatically. Empirically, it is considered that absolute zeta potential values higher than ± 30 mV are indicative of stable dispersions. Only the magnitude of the zeta potential indicates the stability of the sample, whereas the sign of the zeta potential shows whether positive or negative charges are dominant at the surface. Below ± 30 mV processes like aggregation, sedimentation, and/or flocculation are more likely.
The electrochemical double-layer
Surface charge formation
When a solid surface comes in contact with an aqueous medium, charge formation occurs and results in a surface potential Ψ0.Charge formation occurs due to
- reactions of functional groups or
- adsorption of ions from the solution.
Charge formation by reaction of functional groups is based on acidic and basic surface groups. Acidic groups such as carboxylic or sulfonic acid dissociate when in contact with water, i.e. the H+ ion is released into the surrounding water and the surface assumes a negative charge. Basic groups such as amine groups become protonated when in contact with water, i.e. the surface assumes a positive charge (Figure 1). The equilibrium of dissociation and protonation is strongly dependent on the pH value of the liquid medium and therefore has a strong influence on the formed surface charge of the material and further on the zeta potential.
However, the presence of functional groups is not a prerequisite for charge formation. On inert surfaces, negative surface charge is formed due to preferential adsorption of hydroxide ions from water. Inert surfaces are thus negatively charged at neutral and alkaline pH. It is only at low pH, where the concentration of hydronium ions becomes dominant, that inert surfaces exhibit positive surface charge. Again, charge formation by adsorption strongly depends on the pH value (Figure 2).
Immobile and diffuse layer
The above-explained surface charge results in a surface potential Ψ0 and has an effect on the arrangement of anions and cations of the aqueous medium. The charging behavior at the solid-liquid interface and the definition of the zeta potential are well explained using the model of the electrochemical double layer (EDL), which consists of a stationary and a diffuse layer (Figure 3).
- A stationary layer is formed directly at the surface. Ions in this layer are immobilized due to strong interactions with the surface.
- The outer layer is called the diffuse layer because ions are less attracted by the surface and have the ability to move within this layer.
The boundary between the immobilized and diffuse layer is characterized as the shear plane and specifies which part of the electrochemical double layer contributes to the overall net charge of the material. Therefore, the potential which is present at the shear plane is defined as the zeta potential.
Measurement of the zeta potential
Electrokinetic effects like the zeta potential can be determined with four different measurement techniques:
- Streaming potential
- Electrophoresis
- Electroosmosis
- Sedimentation potential
Streaming potential and electrophoresis are the two most common techniques and are implemented in instruments like the SurPASS (streaming potential) and Litesizer (electrophoresis). Streaming potential is typically used for the determination of the zeta potential of larger surfaces (diameter >25 µm) and electrophoretic methods like electrophoretic light scattering are applied for measuring the zeta potential of particles with a diameter of up to 100 µm.
Streaming potential
Measurements of the streaming potential are applied for macroscopic surfaces, where samples are mounted on sample holders and form a capillary flow channel (Figure 4). Upon relative movement of the liquid with respect to the solid sample, the ions of the electrochemical double-layer are sheared off their equilibrium position and shifted along the solid surface. The resulting charge separation gives rise to electrokinetic effects, one of these is called streaming potential. Streaming potential, or alternatively streaming current data, are used to calculate the zeta potential.
The fundamental equations that relate the streaming potential and the streaming current to the zeta potential have been derived by Hermann von Helmholtz and Marjan von Smoluchowski.
The equation used for the calculation of zeta potential using streaming current data requires exact knowledge about the length and cross-section of the streaming channel, i.e. solid sample size.
Equation 1:
dl/dp: slope of streaming current vs. differential pressure
η: electrolyte viscosity
ε: dielectric coefficient of electrolyte
ε0: permittivity
L: length of the streaming channel
A: cross-section of the streaming channel
The equation is thus well suited for zeta potential investigations of planar solids, but is not suitable for the zeta potential evaluation of irregularly shaped samples.
For sample types where the geometry of the streaming channel is unknown (e.g. planar solids of irregular size, fibers, textiles, and granular samples) a derivative of the Helmholtz-Smoluchowski equation is applicable. This equation uses streaming potential data in combination with the electrolyte conductivity.
Electrophoretic light scattering (ELS)
The zeta potential of particles is typically measured by Electrophoretic Light Scattering (ELS). In contrast to streaming potential measurements no movement of the liquid is generated, but the movement of the particles is induced. Therefore, an electric field is applied and the electrophoretic mobility of particles is used to calculate the zeta potential. Due to the electric field particles will move at different speeds: highly charged particles will move faster than less charged particles.
The electrophoretic mobility (speed of the particles within an electric field) is determined through Phase Analysis Light Scattering (PALS). The movement of the particles within the applied electric field results in a frequency shift of an incident laser beam, also known as Doppler shift. The recorded frequency shifts are proportional to the speed of the particles and can be used to measure the electrophoretic mobility and the zeta potential.
However, the actual frequency shifts observed in ELS are too small to be measured directly. Further, they only provide information about the absolute value of the zeta potential, but not about the sign (whether the value is positive or negative). These limitations are solved by using a reference beam, which is then combined with the measurement signal again at the scattering detector (Figure 5). The reference beam is Doppler-shifted by a modulator resulting in a frequency shift, which needs to be known for the calculation of the measurement result. The patented cmPALS technique (continuously monitored phase analysis light scattering) uses an additional detector for live monitoring of the Doppler shift of the modulated reference beam (monitor detector in Figure 5) instead of a theoretical calculation. This results in faster measurements, higher reproducibility even at low applied electric fields as well as higher sensitivity of the measurement.
ELS measurements are used for particle analysis and the method is often combined with Dynamic Light scattering, which is used for particle size determination based on particle movement due to Brownian motion.
Zeta potential and its dependences
The zeta potential depends on both the surface of the sample itself but also the properties of the liquid phase. The pH dependence of zeta potential is among the most extensively studied dependences of zeta potential (explained above). Measurements at different pH values give valuable information on the composition of the sample’s surface, i.e. the presence of acidic or basic functional groups. Automated systems for pH-dependent measurements thereby prevent repeated long-lasting manual adjustments of the pH value. The pH value at which the zeta potential is 0 mV is known as the isoelectric point and is used as an indicator for the chemistry of a surface and is therefore used for the characterization of proteins and peptides as well as for the development of new pharmaceutical formulations.
Besides the pH value, the concentration of ions in the aqueous phase (e.g. through different buffer concentration) also has an influence on the zeta potential. If more ions are available the initial surface charge can be compensated much faster, which results in a minor zeta potential. That means, by increasing the buffer concentration the zeta potential decreases. This effect can be observed e.g. through dilution with pure water instead of the initial solvent.
Zeta potential for adsorption studies
The zeta potential is sensitive to the outermost surface layer of the material. It is thus perfectly suited for monitoring changes in the surface charge upon adsorption of dissolved substances in solution on the solid surface. Both the time- and concentration dependence of adsorption processes are directly accessible by monitoring the respective change in either streaming potential, streaming current, or zeta potential data.
Knowledge about the surface charge and its changes due to liquid-on-solid surface adsorption is important for tuning material properties and optimizing processes. Changes of surface properties due to modification, storage, aging, or wear during operation can be investigated.
Conclusion
Zeta potential measurement is a common characterization method for macroscopic surfaces as well as for particulate systems. Among several techniques, streaming potential and electrophoretic measurements like the cmPALS technique of the Litesizer 500 are the most common.
Learn more about zeta potential analysis on solid surfaces with the Anton Paar SurPASS 3
Learn more about cmPALS and the Litesizer Series for complete particle analysis
Learn more about surface charge and zeta potential measurement systems at Anton Paar
Further Literature
Anton Paar. (n.d.) Faster, More Sensitive Zeta-Potential Measurements with cmPALS and the Litesizer™ 500. [January 24 2019].
Bellmann, C., Caspari, A., Moitzi, C., Fradler, C., Babick, F. (2018) DLS & ELS Guide.
Luxbacher, T. (2014) The Zeta Guide.