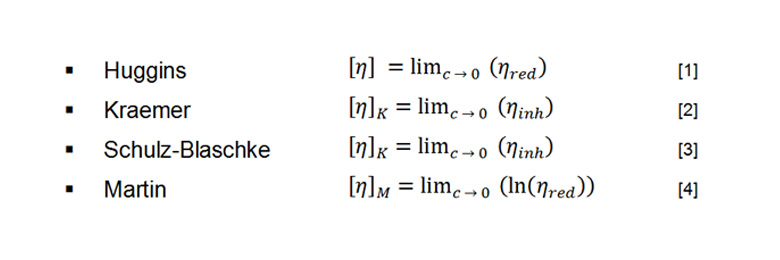Intrinsic viscosity determination – dilute solution viscometry
Intrinsic viscosity determination is used in the field of polymer chemistry, which is a chemistry subdiscipline that deals with the synthesis of polymers as well as the analysis of a polymer’s structure and properties. Intrinsic viscosity is used to classify polymers and helps to identify the applications that specific polymers can be used for.
Polymers are macromolecules that are composed of small repeating units called “monomers” (Figure 1). Depending on the type of monomer as well as the size and molecular structure of the polymer molecule, polymers show unique properties.
When it comes to applications like food packaging, synthetic polymers are mostly used for them because of certain benefits these polymers offer, including versatility, functionality, affordability, and flexibility. Since they are typically derived from fossil raw materials, however, synthetic polymers are increasingly being replaced by biodegradable materials because of environmental concerns. For such materials, these should not only be biodegradable but also be derived from natural resources. To meet this demand for alternative materials for specific applications, many different biopolymers and renewable resource-based biopolymers have been studied and developed. [1]
According to IUPAC, biopolymers are substances that are composed of one type of biomacromolecules (i. e., macromolecules that are formed by living organisms). This definition includes proteins, nucleic acids, and polysaccharides. [2] In comparison to synthetic polymers, biopolymers offer advantages like well-defined and more-complex structures, (bio)degradability, non-toxicity, and renewability [3, 4]. Due to these beneficial properties, biopolymers can be used in different application areas, such as the food, medical, and pharmaceutical industries. More specifically, biocompatible and biodegradable biopolymers are suitable for applications such as edible films, emulsions, or packaging materials in the food industry as well as wound dressing materials, medical implants, sutures, or drug transport materials in the pharmaceutical and medical industries. [1] In addition, they are used as industrial plastics, clothing fabrics, absorbents, water treatment chemicals, or biosensors in other applications. [3]
For the characterization of synthetic polymers as well as biopolymers, many different techniques are available. One of those is “dilute solution viscometry,” which can determine parameters such as intrinsic viscosity.
Determination of the intrinsic viscosity
Dilute solution viscometry
Dilute solution viscometry is a well-known analytical technique in polymer chemistry for characterizing polymers in solution. The technique is based on the fact that dissolving a polymer in a solvent increases the viscosity of the final polymer solution. The increase of viscosity depends on the temperature, the type of polymer and solvent, the concentration of the polymer, and the molar mass of the polymer. Generally, it can be said that the higher the molar mass of a polymer, the higher the viscosity of the polymer solution will be.[1]
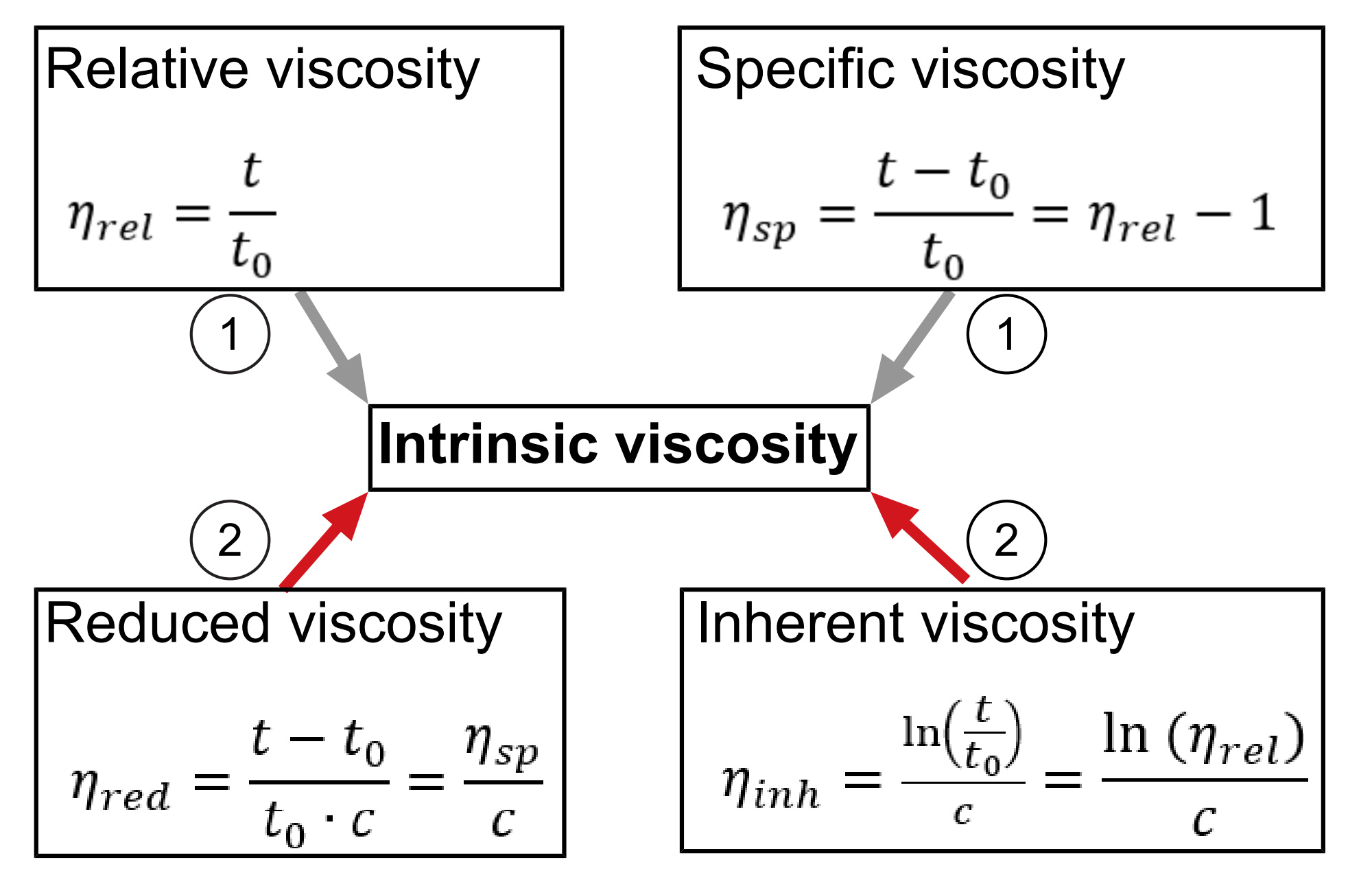
The viscosity of the pure solvent and the viscosity of the polymer solution (polymer dissolved in solvent) are set in relation to each other, which leads to a relative viscosity value. From this basic parameter various polymer parameters can be calculated as shown in Figure 2[1].
The flow behavior of the polymer solution is highly dependent on the molecular structure of the polymer as well as on interactions of the molecules with each other in solution. As the molecular interactions decrease with decreasing concentrations, viscosity measurements are carried out with very dilute solutions. A solution free of any interactions between the polymer molecules could only be reached in the state of the “ideal dilute solution”.[4] In this “ideal dilute solution” the concentration approaches zero meaning that the polymer molecules are isolated from each other and only interact with the solvent molecules. However, this state can never be reached in reality and therefore small polymer interactions have to be considered.[4] Both the calculation of the reduced and the inherent viscosity require the concentration value and therefore the intrinsic viscosity is an important parameter as it is an extrapolation to a theoretical zero concentration.
Various types of glass capillary viscometers, especially Ubbelohde-type glass capillary viscometers, are used for the determination of the intrinsic viscosity and other polymer parameters. Falling-ball- and rolling-ball viscometers are suitable alternatives. The rolling-ball viscometer in particular offers several advantages in comparison to Ubbelohde viscometers . It has a smaller footprint and energy consumption as well as lower solvent and sample consumption. Furthermore, the system is closed and can be automated which makes handling much safer, efficient, and convenient for the users.
Independent of the instrument used, the determination of the intrinsic viscosity can be done in two ways as described in the following paragraphs.
Single-concentration measurements
Multi-concentration measurements are very time-consuming as first the sample preparation for various polymer solutions has to be done followed by viscosity determination of the various polymer solutions.[4] Therefore, it is possible to determine the intrinsic viscosity from one single polymer solution as well, especially when the intrinsic viscosity is only determined approximately. Most of the equations used are simplifications of the equations used for multi-concentration measurements and derive from the relative and specific viscosity (Figure 3).[6]
Multi-concentration measurements
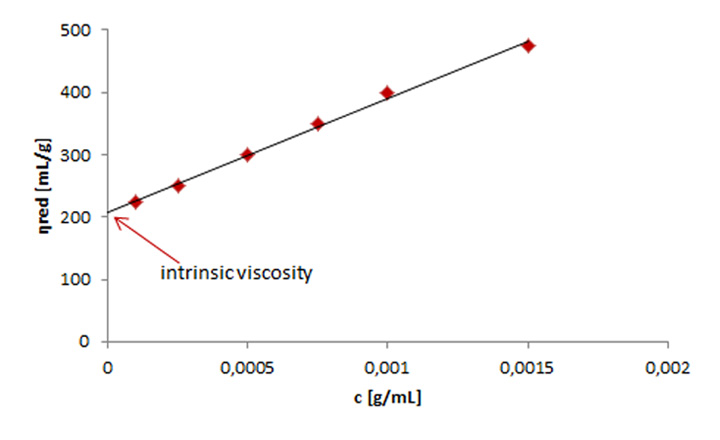
In multi-concentration measurements polymer solutions with different concentrations are prepared.Depending on the regression afterwards, the specific, reduced, or inherent viscosity is determined from these solutions.[2] The determined viscosity is then plotted against the concentration of the investigated polymer solutions.[1] The intersection with the y-axis gives the intrinsic viscosity. To guarantee an exact analysis the relative viscosities of the investigated polymer solutions should lie between 1.2 and 2.5.[4] Various regression models can be used, depending on the type of polymer investigated.[2][4] Some exemplary regressions are listed in Figure 4.
Before use, it has to be tested which equation is applicable for a certain polymer-solvent combination. As an example an extrapolation of the reduced viscosity to zero concentration using the Huggins equation (Figure 4, [1]) is shown in Figure 5.
Intrinsic viscosity calculation
The intrinsic viscosity represents the most relevant variable for describing the viscous behavior of a polymer solution. This is due to the fact that intrinsic viscosity gives the true viscosity-enhancing properties of a polymer independent of its concentration in solution.[2] It is used in various industries to describe the storage stability and quality of a product. One example in which the intrinsic viscosity is used is the plastics industry. However, intrinsic viscosity is also a relevant testing parameter in the pharmaceutical industry (e. g., for the molar mass determination of products such as sodium hyaluronate (hyaluronic acid). As such it is listed in several monographs of the EU Pharmacopoeia and US Pharmacopeia.
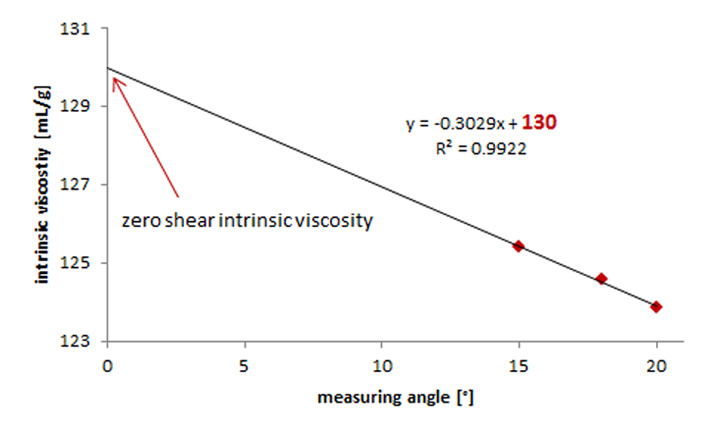
Viscosity measurements for calculating the intrinsic viscosity should always be done at a shear rate approaching 0. At high shear rates the viscosity becomes shear-dependent as the polymer solutions start to show non-Newtonian behavior which is not the case at low shear rates (zero-shear viscosity).[4] To determine the intrinsic viscosity at a shear rate approaching zero, polymer solutions can be measured at various shear rates. The viscosity values obtained are then extrapolated. The intersection with the y-axis gives the zero-shear intrinsic viscosity (Figure 6).
Intrinsic viscosity and molar mass
The intrinsic viscosity of a polymer in a certain solvent can be correlated with the molar mass via the Mark-Houwink equation.[3]
$$[\eta] = KM^{a}$$
$[\eta]$ = intrinsic viscosity
K,a = Mark-Houwink constants
Figure 7: Mark-Houwink equation
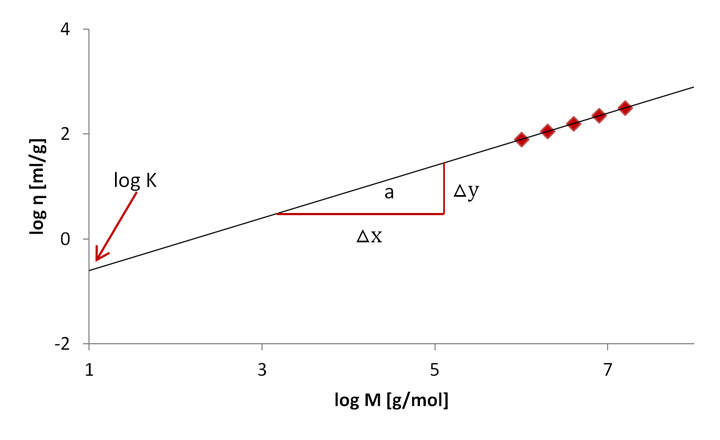
K and a are constants for a given polymer-solvent combination at a certain temperature. If they are not known from literature, they have to be determined experimentally by plotting the logarithm of the intrinsic viscosity as a function of the logarithmic molar mass (Figure 8). The intersection with the y-axis of a linear fit of data gives the constant log K, the slope of the curve gives the constant a (△x/△y).[4]
Examples of K and a for some polymer-solvent combinations are given in Table 1.
| Polymer/Solvent | Temperature | K x10^5 [dL/g] | a |
|---|---|---|---|
| Polypropylene/Cyclohexane | 25 °C | 3.97 | 0.820 |
| Cellulose Acetate/Water | 25 °C | 20.9 | 0.600 |
| Hyaluronic Acid/0.2 M NaCl | 25 °C | 22.8 | 0.816 |
| Polystyrene/Toluene | 20 °C | 11.2 | 0.722 |
| Natural rubber/THF | 25 °C | 10.9 | 0.790 |
Conclusion
The intrinsic viscosity is one of the most frequently determined parameters used to characterize polymers. It is used in a wide range of applications, from quality control in plastic processing and plastic recycling to quaility control of ingredients for the production of medicine in the pharmaceutical industry. Among other analytical techniques “dilute solution viscometry” is widely used in polymer chemistry as it is cheap, easily accessible, but also precise. Therefore, it is present in many laboratories around the world and several ISO and ASTM standards are available for testing a large variety of polymers.
References
- Baranwal, J., Barse, B., Fais, A., Delogu, G. L., Kumar, A. 2022. “Biopolymer: A Sustainable Material for Food and Medical Applications.” Polymers 14, no. 5: 983. doi.org/10.3390/polym14050983.
- Vert, M., Doi, Y., Hess, M. et al. 2012. „Terminology for biorelated polymers and applications (IUPAC Recommendations 2012).” Pure Appl. Chem. 84, no. 2: 377–410. http://dx.doi.org/10.1351/PAC-REC-10-12-04.
- Rebelo, R., Fernandes, M., Fangueiro, R. 2017. „Biopolymers in Medical Implants: A Brief Review.” Procedia Engineering 200: 236–243. doi.org/10.1016/j.proeng.2017.07.034.
- Bibire, T., Yilmaz, O., Ghiciuc, C. M. et al. 2022. “Biopolymers for Surgical Applications.” Coatings 12: 211. doi.org/10.3390/coatings12020211