Zeta Potential III: Surface zeta potential in industrial application
This article gives you an overview of the benefits of zeta potential analysis for various industrial applications. It can help characterize, modify, optimize, and monitor various surface properties.
Zeta potential analysis has become an important characterization method for solid surfaces. Why? Because getting information on the surface charge improves developmental research and quality control for various application fields, including for food preservation, textile production, biomaterial development as well as polymer, membrane, and coating characterization. Zeta potential values change with the pH. Determination of the isoelectric point (IEP), which represents the pH value where the surface charge is reversed and the zeta potential is 0 mV, gives you the opportunity to characterize your sample or modified surface in more detail.
To conduct zeta potential analysis on macroscopic solids, SurPASS 3 is the instrument of choice. It combines very high measurement sensitivity – helping it obtain reproducible zeta potential results in the range of a few mV – with a large variety of measurement cells adapted to a broad range of sample geometries. An integrated titration unit enables a fully automated pH scan. As an additional feature, the instrument is capable of recording liquid-on-solid surface adsorption and desorption kinetics.
Textiles
Certain processes (e.g., dyeing, along with brightness and finishing operations) are strongly related to textile surface properties and to interactions with the environment. You can use the surface zeta potential to monitor and characterize the functionalizing of textiles – which is done to create impregnated wipes for hygiene or bactericidal and insecticidal curtains – and as an indicator for the cleaning effectiveness of laundry detergents and softeners.
For laundry to be successfully washed, textiles have to adsorb detergents and fabric softeners. Adsorption and desorption processes are strictly related to the interfacial interactions between the aqueous bath and the textile (1). Zeta potential change can be used to monitor these adsorption and desorption kinetics.
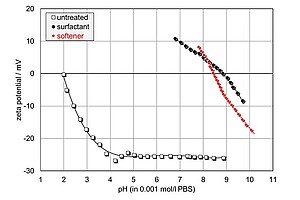
The graph displays the zeta potential over pH of an untreated textile blend composed of 70% cotton and 30% modacrylic fibre before and after adsorption of a quaternary ammonium (quat) or a softener. After adsorption of detergents (quaternary ammonium, softener), the IEP shifts from an initially acidic pH towards a basic pH. This indicates a change of the chemical behavior of the fabric after adsorption.
Polymers
Although you can find polymers being used in various industrial applications, their inert surface can be a constraint. Modifying or activating the polymer surface prior to further processing, however, lets you enhance a material’s wettability, printability, or adhesion. Surface zeta potential measurements can correlate changes in the charging behavior at the solid-liquid interface with the degree of surface activation. This gives you a quantitative view of surface modifications and a reproducible quality of products.
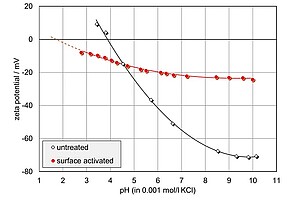
The graph shows low-density polyethylene (LDPE) foils that were surface-activated by UV light irradiation and exposed to sulfur dioxide (SO2). The graph compares the pH-dependent zeta potential of the untreated foil with a surface-activated one. You can see significant changes in both the IEP and the zeta potential. The inert surface of the untreated foil marked by an IEP at pH 4 changes dramatically. The IEP shifts to pH 2, and the final absolute zeta potential decreases in magnitude indicating a chemically modified surface.
Medical devices
In healthcare, medical devices are everyday items for administrating drugs and life-saving treatments. It’s vital that syringes, catheters, heart valves or stents, and many other instruments, perform well. Surface characterization and modification (e.g., coatings) can optimize how you use medical devices. Zeta potential measurements help you characterize surface properties, evaluate coatings and cleaning efficiencies, and monitor how well bioactive substances or components are adsorbed.
Take the example of syringes. They are either prefilled or empty and are used every day to administer particular drugs. The inner surface of the syringe is in contact with the applied substance. Adsorption and any reactivity with the inner surface need to be avoided in order to guarantee efficiency and safety of the applied substance.
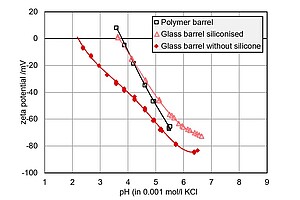
This graph shows the comparison of the pH-dependent zeta potential of polymer and glass syringe barrels. Here, siliconized glass barrels behave very similarly to polymer barrels, with an IEP at pH 4 and a highly negative zeta potential in the acidic range. For glass barrels without silicone coating, the IEP shifts to pH 2.3, revealing the efficient removal of the silicone layer.
Semiconductors
Since the zeta potential results that are determined from steaming current measurements are unaffected by the material’s surface conductivity, these are suitable for the surface analysis of conductive samples. Surface zeta potential has many applications in the semiconductor processing field, including helping you characterize the self-assembly of ultrathin films and the effect of different etching strategies for wafers. It can also help predict particle-wafer interaction in its practical application in the chemical mechanical polishing (CMP) process.
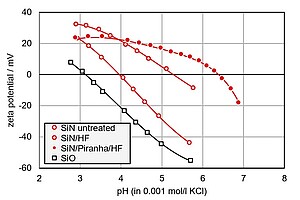
To test the cleaning efficiency, a silicon wafer with a silicon nitride top layer was exposed to different etching protocols. This is shown in the graph. The pH dependence of the zeta potential clearly shows the difference between the as-received silicon nitride wafer (SiN untreated) and a silicon wafer with native silicon oxide coating (SiO). When rinsed with diluted hydrofluoric acid (HF), the isoelectric point of the silicon nitride wafer gets shifted towards a higher pH. Treating it with hydrofluoric acid removes the partial oxidation layer that develops on the surface of silicon nitride during storage. Oxidation with Piranha solution prior to the rinse with hydrofluoric acid shifts the isoelectric point even further towards a higher pH. This treatment is used to remove organic contaminants that protect the as-received silicon nitride wafer from attack by hydrofluoric acid. Surface zeta potential thus helps monitor the effect of different cleaning strategies
Biomaterials
Biomaterials are substances or materials that have been engineered to interact with biological systems. They are designed for medical or therapeutic purposes such as replacing a tissue function or a piece of bone in the body. Due to this, biocompatible materials need to be engineered and optimized. Here, surface characterization and modification both play major roles in minimizing the health risks of various products (e.g., implants). The adsorption kinetics of proteins on an implant’s surface are the first step for cell growth – and thus biocompatibility – making their analysis of major interest as well
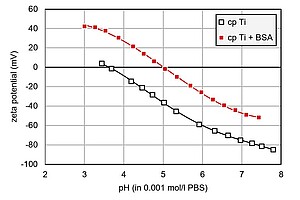
A typical biomaterial is titanium, which is used to make artificial joints and dental implants. The graph shows a comparison of the pH-dependent zeta potential of untreated and coated titanium discs. The untreated titanium (cp Ti) has an IEP of 3.6 and a highly negative absolute zeta potential in the physiological pH. When exposed to a protein solution of bovine serum albumin (cp Ti+BSA), however, the titanium surface attracts this globular protein, and the zeta potential reveals the changes in the surface charge. Here, you can see a significant shift in the IEP to 5 and a decrease in the absolute zeta potential magnitude, which demonstrates what an efficient coating can do.
Cosmetics
Cosmetics change the surface properties of hair and skin. Throughout the day, both of these are exposed to water, light, heat, and even pollutants. Hair needs to be cleaned and softened without destroying its structure or irritating the scalp. Monitoring these processes with surface zeta potential studies based on streaming potential technology lets you evaluate the efficiency and quality of cleaning reagents and cosmetic products.
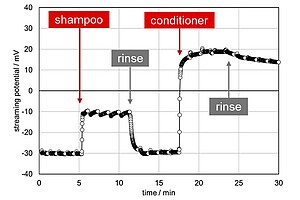
Shampoo, conditioner, and rinsing cycles all influence hair. By determining the streaming potential of these, you can monitor their influence over time.
Applying shampoo decreases the absolute streaming potential, which reveals the change in surface chemistry of the hair sample. This is reversed with a rinsing cycle. In contrast, applying conditioner decreases the streaming potential magnitude and reverses the charge. This change is not reversed by rinsing, indicating the conditioner’s excellent efficiency.
Filtration and membranes
Membranes are selective barriers that allow a subset of particles to pass through while stopping others from doing so. Since they separate particles, they are used in several application fields, including in the food and chemical industries, biotechnology, pharmacy, and wastewater treatment.
Surface charges, determined by zeta potential measurements, are a key parameter that describe the interaction between the membrane and its environment. This information lets you optimize and monitor membrane filtration processes.
References
- Luxbacher, T. & Juriniak, Ana & Čurlin, Mirjana & Petrinic, Irena & Buksek, Hermina & Pusic, Tanja. (2014). Assessing the rate of adsorption of a softener on knitted fabrics by streaming potential measurement