BET Theory
The BET theory (abbreviated from Brunauer-Emmett-Teller theory) is used to measure the surface area of solid or porous materials. It gives important information on their physical structure as the area of a material’s surface affects how that solid will interact with its environment. Many properties such as dissolution rates, catalytic activity, moisture retention, and shelf life are often correlated to a material’s surface area. Critical to the design and manufacture of solids, surface area analysis is one of the most widely used methods in material characterization. This article gives insight into the procedure of BET surface analysis and its application in industry.
BET surface area determination: measurement principle
The surface area of a material can be altered during both synthesis and processing. As a particle is divided (milled) into smaller particles, additional surfaces are created, thereby increasing its surface area. Similarly, when pores are created within the particle’s interior, by dissolution, decomposition, or some ther physical or chemical means, the surface area is also increased. Materials with many narrow pores, such as activated carbons, can have surface areas of more than 2,000 m2 in a single gram.
Using the BET theory, the true or specific surface area, including surface irregularities and pore walls, of a particle is determined at an atomic level by adsorption of an unreactive gas. Because most gases and solids interact weakly, the solid material must be cooled, typically using a cryogenic liquid. The temperature of the solid sample is kept constant, or under isothermal conditions, while the pressure or concentration of the adsorbing gas is increased. Figure 1 depicts a typical surface area isotherm, where the x-axis is the relative pressure of the gas and the y-axis is its volume adsorbed onto the sample.
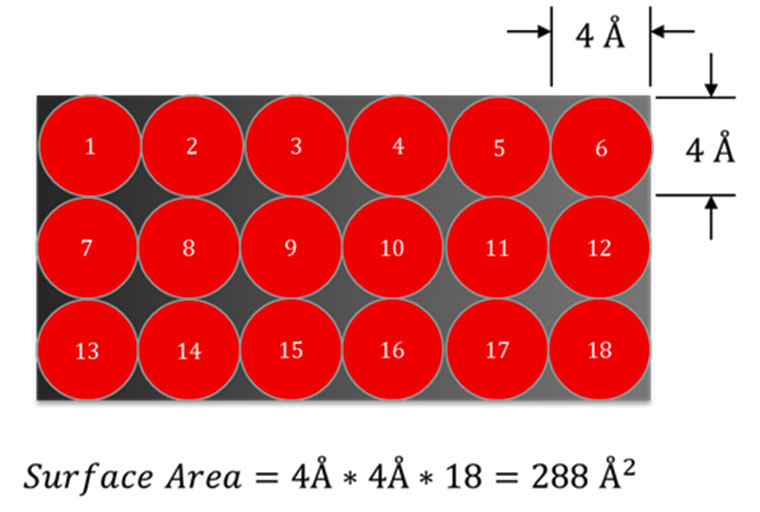
As the relative pressure is increased, more and more molecules adsorb on the surface. A thin layer will eventually cover the entire surface (dark red in Figure 2) in a single or mono-layer. The number of gas molecules in the monolayer is recorded from the volume adsorbed. Since the cross-sectional area of the adsorbate is known, the area of the accessible surface may be calculated. However, gas adsorption as a function of pressure does not follow a simple linear relationship (as shown in Figure 1). Therefore, an appropriate mathematical model must be used to calculate the surface area: the BET equation.
BET equation
The BET equation (named after Brunauer, Emmett, and Teller who developed the theory) was first published in 1938 (Brunauer, 1938), and continues to be the most widely used method to determine the number of molecules/atoms of a gas needed to form a monolayer, Xm, of adsorbed gas on a solid surface (Thommes, 2015). The BET equation (Equation 1) describes the relationship between the number of gas molecules adsorbed (X) at a given relative pressure (P/P0), where C is a second parameter related to the heat of adsorption.
The BET equation strictly describes a linear plot of 1/[X(P0/P)-1] vs. P/P0 which for most solids, using nitrogen as the adsorbate, is restricted to a limited region of the adsorption isotherm, usually in the P/P0 range of 0.05 to 0.35. A typical BET plot is shown in Figure 4.
The surface area, SA, is calculated from the slope and intercept according to Equation 2
where CSA is the cross-sectional area of the adsorbate (Lowell, 2004).
Applications of the BET principle
Any solid material may be characterized using gas adsorption for the determination of its surface area. Common applications in which knowledge of the surface area is critical include the production and further processing of carbon, pharmaceuticals, catalysts, batteries, ceramics, and minerals.
Carbon Black
One of the most widely used reinforcing materials in the production of rubber, the surface area of carbon black impacts its performance in different applications. Higher surface areas are used for high wear resistance applications (tire treads), whereas lower surface area carbon blacks are characterized by low hardness and tensile strength (and therefore well-suited for use as wire insulation, for example).
Pharmaceuticals
Catalysts
Heterogeneous catalysts, primarily solids, are used in many industrial chemical processes and are typically comprised of a reactive species on a non-reactive or inert support. The surface area of both components influences the rate and yield of the reaction, which makes it of paramount importance to researchers and manufacturers alike.
Batteries
The performance of various battery components, such as anodes, cathodes, and separator membranes, may be affected by their surface areas. Properties such as charging and discharging rates, impedance, and capacity are related to the surface areas of these materials. For more information, see the application report "Surface Area Determination of Battery Cathode and Anode Materials".
Ceramics
Ceramics are used in the manufacture of thousands of everyday items including general purpose products like glass and cement as well as more technical products such as semiconductors and microchips. The surface area has an impact on sintering, thermal properties, and moisture retention, and thus is an important property in the selection and production of ceramics.
Conclusion
As the BET principle employs gas adsorption data it is equally applicable to porous and non-porous materials regardless of particle size and shape. As such, it is a useful tool for the investigation and manufacture of a wide variety of solids.
References
Brunauer, S. et al. (1938). Adsorption of Gases in Multimolecular Layers. Journal of the American Chemical Society, pp. 309–319.
Lowell, S. et al. (2004). Characterization of Porous Solids and Powders: Surface Area, Pore Size and Density. 1st ed. Dordrecht, The Netherlands: Springer.
Thommes, M. et al. (2015). Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure and Applied Chemistry, pp. 1051–1069.