Biological nanomaterials

When talking about biological nanomaterials, we typically think of nanoparticles. But other kinds of nanomaterials have also proved very useful for applications like drug delivery, regenerative medicine, and many more. In this section, you will find a research case that describes the structural characterization of biological macromolecules in solution with SEC-SAXS. Furthermore, you will find articles on dynamic and electrophoretic light scattering of virus and virus-like particles and exosomes, as well as viscosity testing of biomaterials for nanomedicine. All these techniques are very valuable for research on biological nanomaterials. Anton Paar offers not only the instruments for the techniques, but also valuable support and expertise – to drive your research forward.
SEC-SAXS – Structural characterization of biological macromolecules in solution
Introduction
Molecular interactions are important for the function of biological macromolecular assemblies and systems. Small-angle X-ray scattering (SAXS) can provide key structural and dynamic information for biological macromolecules such as proteins, and nucleic acids and their complexes in solution, under near-native conditions. A prerequisite for accurate interpretation of solution SAXS data is that the macromolecule studied is present in monodisperse form. Only then is it possible to derive the 3-dimensional structure of proteins in high quality. In practice, many samples comprise aggregated structures and oligomeric mixtures, respectively, which complicate a precise evaluation of scattering data. To overcome this problem, SAXS can be coupled with size exclusion chromatography (SEC) and thus provides an efficient tool for screening and structural studies of biomolecular samples. SEC – which often is the final step in many protein purification protocols – separates biomolecules based on their hydrodynamic properties (size, shape). The method is specific enough to separate even different oligomeric species, such as monomers, dimers, and higher oligomers.
Experimental
The capabilities of combined SEC-SAXS were demonstrated by separating and analyzing complex mixed protein solutions of human serum albumin (HSA) and lysozyme by in-line as well as offline coupling. An Äkta Pure 25 FPLC system was used to separate the protein mixtures, while SAXS data of the eluted protein fractions were measured with an Anton Paar SAXSpace system. For automatic processing of the measured SEC-SAXS data, principle component analysis (PCA) and partial-least-squares discriminative analysis (PLS-DA) were used. PCA and PLS-DA are multivariate statistical techniques allowing automatic analysis of complex protein samples imposing several challenges, e.g. varying concentrations, varying biomolecular species over the SEC run, and potential co-elution of different biomolecules.
Results and discussion
Complex protein mixtures were successfully separated and measured using SEC-SAXS with laboratory instrumentation, resulting in effective separation of the different protein fractions. The measured scattering data were evaluated using a PCA and PLS-DA. Notably, peaks of closely eluted fractions and of different biomolecules could be automatically discriminated, without the requirement of manual data analysis. Therefore, even complex protein samples comprising large aggregates as well as mixtures of different oligomers could be efficiently analyzed.


Additional information
Instruments:
Application reports:
Investigation of inactivated virus and virus-like particles
Introduction
Most antiviral vaccines consist of inactivated virus, recombinant proteins or virus-like particles which have no potential for replication. Many of them are administered together with vaccine adjuvants, which increase the efficacy and longevity of the immune response. The oldest and still most widely used adjuvant is aluminum salt (e.g. aluminum hydroxide). The immunogenicity of the vaccine is strongly related to the particle size. Viruses are nanoparticles ranging in size from 15 to 300 nm. Upon injection, particles in this size range are efficiently taken up by dendritic cells, a class of sentinel cells uniquely endowed with the ability to induce both antibody- and killer cell-mediated immunity. Aluminum salts, in contrast, are in the micrometer range and are preferentially taken up by monocytes and macrophages, which predominantly induce an antibody-mediated immune response. Therefore, the particle size of the vaccine needs to be monitored, and tailored based on the type of immune response required to counter the pathogen. The zeta potential gives additional information for the quality control of vaccines preparations.
Experimental
Samples: Inactivated tick-borne encephalitis (TBE) vaccine, based on the Neudörfl strain and adjuvanted with aluminium hydroxide. Inactivated quadrivalent influenza vaccine, containing 2 strains of influenza A viruses and 2 strains of influenza B viruses according to the WHO guidelines for the 2019-2020 northern hemisphere influenza season.
Sample treatment: The vaccines were measured after continuous storage at +4 °C as well as after conditioning at 50 °C (“heat-treated”) or at -18 °C (“freeze-thawed”) for 24 h.
Characterization technique: The samples were characterized via particle size and zeta potential with the Litesizer 500.
Results and discussion
TBE vaccine stored at 4 °C displays a single peak culminating between 2 µm and 3 µm corresponding to the particle size of commercial aluminum hydroxide used as vaccine adjuvant. A significant increase in HDD is observed for both heat-treated and freeze-thawed vaccine samples, indicating that major disruptions in the cold chain promote adjuvant aggregation. The influenza vaccine shows a bimodal distribution and the freeze-thawing tends to increase the aggregation of split virus particles, while heat treatment reduces it. Both vaccines show a slightly negative zeta potential (low magnitude) which, on the one hand, might promote uptake by sentinel cells, but might also explain their tendency to aggregate in response to cold chain disruptions.
Parameter | Untreated | Heat-treated | Freeze-thawed |
Mean ZP | - 6.6 mV | - 6.0 mV | - 5.7 mV |
Standard Deviation | 0.9 mV | 0.8 mV | 1.3 mV |
Rel. standard deviation | 13.7 % | 12.5 % | 22.4 % |


Additional information
Instruments:
Application report:
Exosomes as new frontiers in the drug delivery
Introduction
Exosomes are nanometer-range vesicles which can be isolated from cell culture medium as well as from many extracellular fluids such as blood, urine, saliva, breast milk and cerebrospinal fluid. Their potential as drug delivery system has been known for years. Their many advantages include a long-range cell-targeting action, low toxicity, low immunogenicity, high stability and the capacity to encapsulate proteins, drugs or nucleic acids. For this reason, exosomes are regarded as new frontiers in the development of vaccines and cancer-targeting therapy. As shown in this report, the characterization of exosomes via particle size as well as the zeta potential allows to monitor the stability, optimize storage conditions and estimate the uptake process from the target cells.
Experimental
Sample preparation: The cell culture medium used for the human cell line TR146, a squamous cell carcinoma originating from the buccal mucosa (from Cancer Research UK), was aspirated and centrifuged. The exosomes were isolated with the Invitrogen™ Total Exosome Isolation kit (Thermo Fisher Scientific). Samples were incubated in the fridge overnight then centrifuged at 10 000 g for 60 min at 4 °C. The resulting supernatant was discarded and the exosome pellet was resuspended in 100 µL PBS Buffer (pH = 7.4, Thermo Fisher Scientific).
Characterization technique: The particle size and zeta potential were measured with the Litesizer 500.
Results and discussion
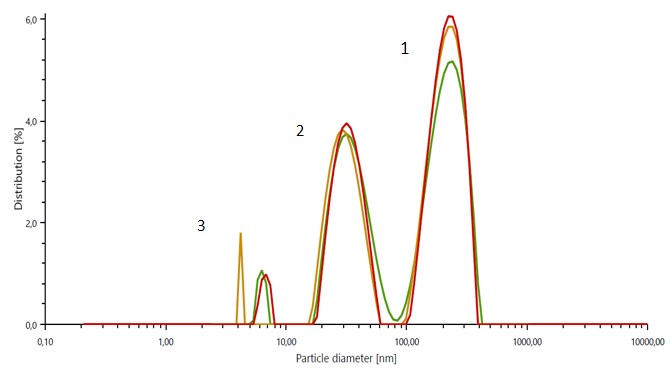
The particle size distribution results of the untreated exosome suspension displayed 3 well-defined peaks, with a peak of large particles (~ 200 nm) corresponding to cell debris, a peak of medium-size particles (~ 30 nm) corresponding to exosomes, and a discrete peak of very small particles (~ 2 -10 nm) indicating the presence of free-floating proteins. The mean particle size of the exosome peak recorded only minor fluctuations over a week of storage at 4 °C. For samples stored at 37 °C, the particle size of exosomes was already significantly decreased after 72 hours. The zeta potential results provide information about the surface charge of exosomes and therefore about their interaction with biological systems.

Additional information
Instruments:
Application report:
Viscosity testing of biomaterials like bentonite clay for nanomedicine
Introduction
Surface functionalization of materials for controlled interaction with various systems of biological interest is of vital importance; biomaterials undergo nano- and microstructuring for their final usage in nanomedicine.
Experimental
500 mL of bentonite clay were filled into a 600 mL beaker and viscosity-tested using a rotational viscometer. A motorized stand adapter for the rotational viscometer and T-bar spindles was used for the measurement due to the paste-like behavior of the material at rest. A linear speed ramp from 1 rpm to 10 rpm with 4 measurement points was performed to evaluate the flow behavior. The measurement point duration was set to 1 min and 30 sec. The motorized stand adapter created a helical movement of the T-bar spindle in the sample and eliminated the “channeling” problem. Any other spindle which rotates at the same height would create an air channel within such a sample. This would lead to meaningless viscosity values, as only part of the sample is in contact with the spindle.
Results and discussion
As expected, the bentonite clay exhibited shear-thinning flow behavior with a Shear Thinning Index of 8.3519. This means that the sample’s viscosity became lower at higher shear rates. Having the proper shear-thinning flow behavior (and adjusting ingredients accordingly) simulates, e.g., a human swallowing a nanomedicine product. The final flow behavior can be adjusted by increasing the concentration of nanoparticle powder or by adding bentonite clay as a thickening agent, if necessary.
Additional information
Instruments:
Application report: